Managing Varroa Mites in Honey Bee Colonies

Honey bee health remains a primary focus of researchers in response to high annual losses of commercial colonies in the United States and Canada. Although not fully understood, high colony mortalities stem from multiple factors that include the parasitic mite Varroa destructor Anderson & Trueman, viruses vectored to bees by varroa mites, pesticide exposure, residues of agrochemicals in hives, and poor nutrition. Varroa mites and the viruses they vector are currently viewed as the primary killers of honey bees worldwide.
The acaricides used to control varroa mites are an additional threat when sequestered as chemical residues in combs. Chemicals placed in colonies contaminate hive products (beeswax) and may cause sublethal effects on individual bees that reduce overall colony health. Using miticides also results in mite populations with genetic resistance to these chemicals. For these reasons, chemicals to control varroa mites should be used minimally. Integrated pest management (IPM) strategies that slow the growth of mite populations and reduce the need for miticides are key to minimizing chemical use. This publication provides an overview of the biology of varroa mites and IPM approaches that can be used to manage varroa mite populations.
Basic Biology of the Varroa Mite
The varroa mite is the most serious pest of the Western honey bee (Apis mellifera L.) throughout the world. This mite was first described on the Eastern honey bee (Apis cerana F.) from Java, Indonesia, in 1904. The mite was originally called Varroa jacobsoni Oudemans, but research published in 2000 indicated that more than one species of varroa mites existed. It turns out that V. jacobsoni usually does not kill or damage colonies of the Western honey bee, and V. destructor is the species causing most harm to bees. The first reports of varroa mites attacking Western honey bees were in the early 1960s in Asia.
The varroa mite has since become established on every continent, including Australia. Varroa mites spread between continents via commercial transport of bees by beekeepers and by swarms that fly long distances or hitch rides on ships, airplanes, and trucks. Varroa mites move within infected regions by the same transport mechanisms. Within apiaries, varroa mites move with drifting bees, or they hitch rides on robber bees that attack hives weakened by mite parasitism.
Adult female varroa mites are oval and flat, about 1.1 millimeter long and 1.5 millimeter wide. They are tan to reddish-brown and can be seen easily with the unaided eye (Figure 1). Male mites are considerably smaller and are a light tan color. Male mites are not usually seen outside brood cells.
Varroa mites reproduce within brood cells, where they feed upon the developing bee pupae to obtain nourishment for egg production (Figure 2). Mite offspring also feed on the host pupa to obtain nourishment for growth and development. When colonies of bees are raising drone brood, varroa mites prefer to invade drone brood about nine times more than worker brood. This is significant because the development time of drones is a few days longer than workers (24 and 21 days, respectively). The mites can produce more adult daughters when in drone brood, and mite populations can grow faster than when a colony only has worker brood.
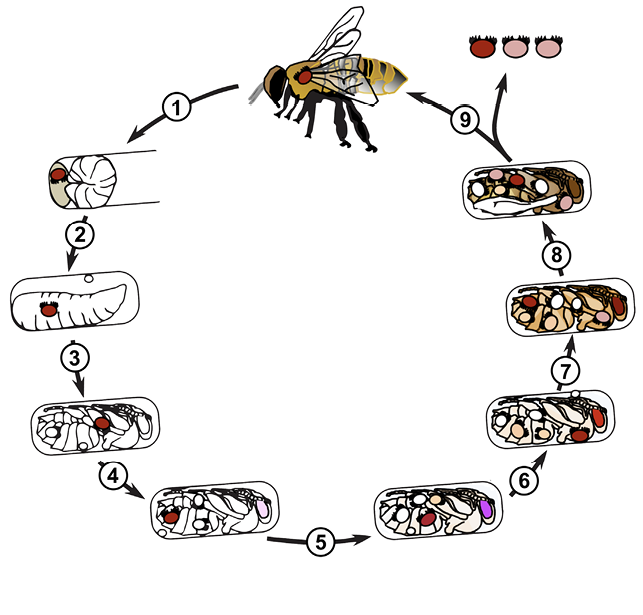
- Adult female mite invades brood cell about 15–18 hours before brood cell is capped.
- The mite lays first egg (male) about 30–35 hours after invading cell.
- The mite lays three to four eggs (female) at about 30-hour intervals.
- Each offspring progresses through two nymph stages before becoming an adult.
- The male mite becomes an adult before his sisters.
- The first daughter matures to adulthood and mates several times with her brother.
- The second daughter reaches adulthood and mates with brother. The oldest daughter continues to darken.
- The bee pupa expands wings and about a day later emerges from the brood cell.
- The mother mite and one to two adult daughters leave a worker brood cell with the emerging bee. Each of these mites lives on adult bees for about a week before invading a new brood cell to reproduce.
Adult bees serve as intermediate hosts when there is no brood available. Varroa mites attach to adult bees between the abdominal segments or between body regions (head, thorax, and abdomen), making them difficult to detect. These are also places from which they can easily feed on the fat body tissues of the adult bee.
Adult female mites invade brood cells that contain 5-day-old worker larvae, and the adult bees cap these brood cells shortly afterward. Each mite lays up to four or five eggs at approximately 30-hour intervals. The first egg usually develops into a male, and the later ones into females. The development of offspring proceeds from egg to six-legged larva (within the egg envelope) to eight-legged protonymph to deutonymph, and finally to a sexually mature adult mite in 5½ days (female) to 6½ days (male) (Figure 3).

The larvae and protonymphs of male and female mites look identical, but the deutonymphs and adults are easy to differentiate. The females are oval in shape, while the males are more triangular. Mites shed their cuticles when molting into the next nymphal stage. However, only the shed cuticles from the last molt (deutonymph into adult mite) are easily seen in brood cells. Young adult mites mate in the capped cells. Adult males and all immature mites die after the host bee emerges from the brood cell (when nest bees clean the cells). Young adult daughters and the mother mite exit the brood cell with the emerging host, and they eventually transfer to other passing bees. A mite enters another brood cell in about 7 days during periods when brood is available. However, varroa mites may survive more than 100 days when no brood is available, such as during the winter months.
Adult bees growing from a mite-infested brood cell suffer from loss of body fat and are often underweight. Their lifespans may be greatly reduced, and the most severe damage occurs when mites vector harmful viruses to the bees. Although many viruses can be vectored, it seems that a particularly virulent form of the honey bee deformed wing virus (DWV) kills most colonies. The degree of damage from direct feeding by the mites depends on the number of mites parasitizing each bee larva. One or two invading mites will produce enough offspring to decrease the vitality of the emerging bee. Higher numbers of varroa mites per cell result in malformations like shortened abdomens, misshapen wings, and deformed legs or even death. Normal colony functions become disrupted when young bees that normally progress through a series of age-related tasks are lost prematurely from the bee population.
Eventually, the mite population increases to economically damaging levels, and the colony weakens and begins to exhibit symptoms of a condition known as parasitic mite syndrome (PMS). The basic symptoms are a very weak colony with brood combs having a poor capped brood pattern and sick larvae showing a yellow or brown color caused by viral diseases (Figure 4). Colonies exhibiting PMS usually cannot be saved.

Integrated Pest Management (IPM) and Varroa Mites
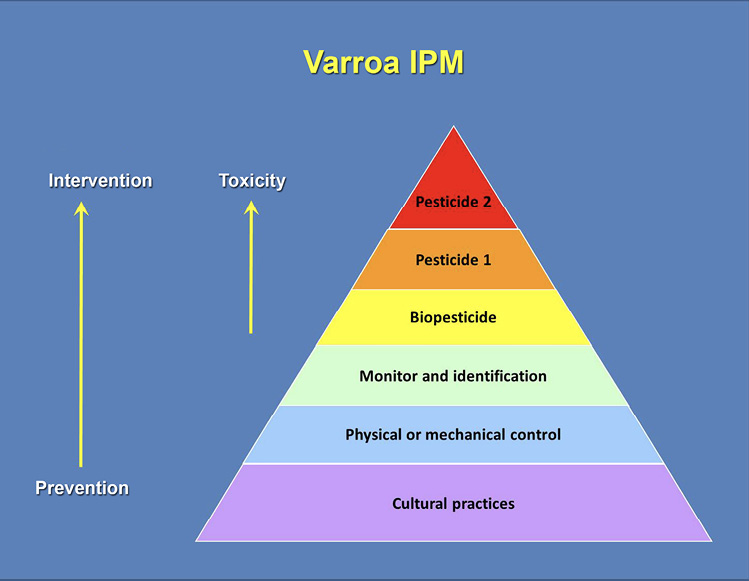
IPM is a method of dealing with pests and parasites that differs from the application of chemical treatments on a regular schedule. It is based on the realization that one cannot chemically eradicate pests or parasites but must continually manage their populations. IPM (Figure 5) involves mixing different tactics (genetic/cultural methods, mechanical/physical methods, and chemical treatments) and knowing critical times in the life cycles of the target organisms that make them vulnerable to control methods. Chemicals are only applied when pest populations are above an action or economic threshold, rather than on a timed schedule. The reduced use of chemicals to control varroa mites limits possible contamination of hive products and delays the development of resistance to the chemical by the mites. It also reduces the exposure of the bees to potentially harmful chemicals. This is especially significant because most of the miticides used to kill varroa mites are known to have some negative health consequences for honey bees.
Sampling for Varroa Mites
A key component of IPM is that decisions to use chemicals for controlling pests are based on sampling (monitoring and identification) the pest population and only treating when a critical threshold has been reached. Generally, the action threshold reflects a population of mites that if not treated could quickly grow to damaging levels that cause economic injury to your hive. The decision on whether or not to apply a chemical is based on thresholds derived from sampling the pest population. In beekeeping, this is not as easy as in row crops, but it is still possible.
The economic threshold for varroa mite populations in colonies with 25,000–34,000 bees is in the range of 3,172–4,261 total mites in August for the southern United States (Delaplane and Hood, 1999). This range corresponds to 9–18 phoretic mites found on a sample of 300 adult bees, which is a mite load of 3–6 percent. Most beekeepers prefer to use the 3 percent end of the range as the action threshold. Migratory beekeepers may need to use a lower threshold because there is a lot of movement of mites between holding yards and among apiaries from different commercial beekeepers at pollination sites. Additionally, research suggests that some viruses vectored by varroa mites are becoming more virulent through time. More research is needed to determine the best minimal thresholds for a variety of beekeeping practices. Until new thresholds are established, it might be prudent to intervene when the total mite load in a colony is 2,000–2,500 mites, which is a population only weeks from a currently accepted economic threshold (Table 1).
|
Time of year |
Total mite population |
Mites on adult bees1 |
Beekeeper action2 |
|
summer |
500 mites |
0.5–0.7% |
Wait; 9–12 weeks for mite population to reach threshold |
|
summer |
1,000 mites |
1.0–1.3% |
Wait; 6–8 weeks for mite population to reach threshold |
|
summer |
2,000 mites |
2.0–2.7% |
Treat with miticide; 2–3 weeks for mite population to reach threshold |
|
autumn |
≥ 2,500 mites |
2.4–3.3% |
Treat with miticide; 1–2 weeks for mite population to reach threshold |
|
anytime |
≥ 3,700 mites |
3.6–5.1% |
Treat with miticide; 1–2 weeks for mite population to reach threshold or at threshold |
1Range given for colony population equal to 24,000–34,000 adult bees.
2Range based on mite population growth rates during typical non-drought seasons in the southern United States.
The mite population is distributed among the adult bees and the capped brood within a colony. Although there is considerable variation, typically one-third of the mite population resides on adult bees and two-thirds are in capped brood. This distribution assumes that brood production is normal and has not been interrupted by the loss of a queen. This assumed distribution is probably less accurate when drone production is highest in early spring, but it provides a reasonable basis for interpreting the different sampling methods to be described below.
In Table 1, the assumed economic or action threshold is 3,700 mites (the midpoint of the range of 3,172–4,261 mites). The mite load on adult bees was calculated by assuming one-third of all mites were on bees (in a bee population of 24,000–34,000 adults). The time to reach 3,700 mites was estimated using exponential growth rates measured for mite populations in a typical spring–summer season (not during hot and dry periods associated with drought) in the southern United States.
Sampling Mites on Adult Bees
The most convenient methods of sampling varroa mites involve estimating the prevalence of mites on adult bees. The prevalence is often described as a percentage. For example, a 3 percent mite load means that there were three adult mites counted for every 100 bees. Most beekeepers prefer not to kill bees during sampling, but people with many colonies might find it more efficient to collect samples from many colonies at once and measure the mite loads at a more convenient time later. The most commonly used nondestructive sampling method is the powdered sugar shake. This method is much less accurate in high humidity conditions when the powdered sugar tends to clump. The clumped sugar is less likely to dislodge mites, and the method will underestimate the mite load on adult bees. The destructive method involves washing samples of dead bees with alcohol or a soapy water mixture. This method is much more accurate than the sugar shake.
Sugar Shake Method
The powdered sugar roll or sugar shake supposedly provides a means for estimating the mite load without killing the adult bees that are sampled. However, research by Dr. Jennifer Tsuruda at the University of Tennessee (personal communication) showed that sugar-coated bees die or are removed from the hive shortly after they are re-introduced into their hive. The method hinges on the ability of dusts with small particle size to dislodge the adult mites from the bodies of the adult bees. The small particles of 10x confectioners sugar create a barrier between the bodies of bees and the specially modified feet of the mites, which are like small suction cups.
The first step is to make a jar for sampling adult bees. A wide-mouthed quart- or pint-sized canning jar works nicely. The trick is to replace the solid dome of the canning jar with a circular piece of 8-by-8 wire mesh cut to fit the jar. The mesh is held in place with the ring normally used to secure the canning dome lid. When bees are rolled and coated in the sugar, the mites’ feet eventually contact the powder and they lose their grip. The sugar can then be poured onto a sheet of wax paper, and the falling mites can be counted. If it is windy, sprinkle the sugar onto a heavy-duty white paper plate that won’t blow away.
To sample a colony, it is best to locate the queen and make sure she does not accidentally end up in the sampled bees. Varroa mites reside on younger bees within the broodnest. Choose two combs with capped and/or uncapped brood from the brood chamber and gently shake the adhering bees from the two combs into an empty box (a cardboard nuc box works well). Mix the bees and then place 400 bees (½ cup) into the sampling jar.
Secure the mesh wire with the canning ring and add 2 tablespoons of powdered sugar through the mesh onto the bees. Mix the bees and sugar to thoroughly coat all of the bees. Wait about 2–3 minutes before turning the jar upside-down and shaking the sugar from the jar onto the wax paper or paper plate. Continue shaking the bees for a minute or so to dislodge all of the mites. You may want to repeat this process to make sure all mites are dislodged from the bees.
The mites will appear as small, brown, oval flecks in the powdered sugar. Sometimes movement of their legs makes them easier to see. If you have trouble seeing the mites, a small amount of water can be added to dissolve the sugar and reveal the mites. To estimate the mite load, count the total number of mites dropped from the sample, divide by the total number of bees that were sampled, and multiply by 100. For example, if you sampled 400 bees and found 20 mites: (20 ÷ 400) x 100 = 5%.
Alcohol Wash
The alcohol wash is similar to the sugar shake except that the adult bees are killed before measuring the mite load. Bees are taken from two combs of capped brood as in the previous section, and 400 worker bees are put into a jar or into a Ziploc bag, which is then sealed. Jars or bags should be labeled to identify each colony being sampled, and samples can be stored on ice during transport from an apiary.
Samples in Ziploc bags can be frozen if washing will be done at a later date. When you are ready to wash frozen samples, transfer the bees from the Ziploc bags into canning jars and add 70 percent ethanol to completely cover the bees. Be sure to examine the Ziploc bags for any mites and transfer them into the jars with the bees.
If bee samples are collected in canning jars while in the field, 70 percent ethanol can be added to them immediately, especially if they are to be washed on the following day or so. The alcohol should completely cover the adult bees. Secure the solid dome lid onto each jar with the ring. Washing works best if the bees have soaked in the alcohol for at least a day.
Shake the bees vigorously for several minutes before pouring them through a filter made of 8-by-8 wire mesh placed over a catch reservoir. The mesh will retain the bees while the ethanol and mites pass into the reservoir (Figure 6). If the reservoir is white, the mites will show up well for counting. After counting the first wash of mites, decant the ethanol from the sample and discard the mites. Wash the sample of bees again. Several washes may be needed to dislodge all mites. The mite load is found as in the previous section by dividing the total number of mites by the total number of bees that were sampled and multiplying by 100.

If you do not have access to ethanol, a soapy water solution can be used to wash the mites from the bees. Add 1–2 milliliters (15–30 drops) liquid dish detergent to 500 milliliters (about 1 pint) of water to make the washing solution. Agitate the bees in the detergent solution for several minutes before filtering. This method is more efficient if you use mechanical shakers for several hours.
Using Mite Drop to the Floor as Indicator of Mite Population
The increased popularity of screen floors in hives as a non-chemical control method for varroa mites (see below) offers a fairly passive and relatively non-disruptive method for sampling colonies. The method is based on the phenomenon that the number of varroa mites falling to the floor correlates to the mite population within the colony. Specifically, the degree of “mite drop” reflects the total mite population—mites in brood cells and on adult bees. Presumably, adult female mites fall from worker bees regularly, and dead adult mites are removed from brood cells and dropped to the floor by nest-cleaning bees. The sugar shake or alcohol wash only estimates the mite load on the adult bees.
The basic procedure is to insert a piece of cardboard covered with adhesive beneath the screened floor of the hive. The 8-by-8 screen mesh allows mites to fall through to the adhesive on the sticky board but prevents the bees from contacting the sampling surface. The adhesive may be a thin layer of non-stick cooking spray or petroleum jelly applied to the board. Many commercial screened bottom boards are made with a groove to which a sticky board can be easily inserted from behind the hive. The commercial sticky boards usually come with a sampling grid to make counting the mites easier. Sticky boards can also be made from white cardboard and cut to fit the typical bottom board.
Leave the sticky board in place for 3 days, and then count the number of dead mites. The daily “mite drop” is estimated by dividing the total mites by the number of days for the sampling period. The economic thresholds estimated for using this technique vary widely from region to region. However, a reasonably conservative threshold for late summer in Mississippi would be a daily mite drop greater than 40–50 mites per day.
Making Apiary-Level Treatment Decisions
Practical sampling plans have been developed that beekeepers can use to make treatment decisions for whole apiaries based on samples from a few colonies (Lee et al. 2010). Commercial beekeepers needing to make apiary-level treatment decisions do not have the time or resources to sample every colony they own, which could be hundreds to thousands of colonies. Although sampling schemes were developed to make apiary-level treatment decisions, a beekeeper cannot make a decision about treating all apiaries based on the sampling decision made for one apiary. This is because the mite loads can vary significantly between apiaries within a single commercial operation.
The basic procedure for apiary-level decisions involves averaging the adult bee mite loads taken from a subset of colonies within the apiary—each colony is sampled by an alcohol wash of 300-400 bees (as above). The average mite density of these samples will reflect the average mite load of the entire apiary. If the average mite load exceeds the action threshold, treat the entire apiary with chemicals to control varroa mites.
The number of colonies sampled in an apiary depends on the number of colonies within the apiary. The colonies sampled should be chosen at random and should include the entire geographical range of the apiary (not just concentrated in one area). For example, apiary-level treatment decisions can be made for apiaries with more than 20 colonies by sampling no more than eight colonies with a reasonable level of precision. This means that a beekeeper samples eight different colonies in an apiary of 25 colonies, and only samples eight colonies in another apiary of 60 colonies. The researchers that developed the schemes did recommend fewer samples for smaller apiaries (Table 2).
|
# colonies in apiary |
# sampled |
|
>20 |
8 |
|
20 |
6 |
|
10 |
5 |
|
4 |
3 |
Nonchemical Methods for Slowing Growth of Mite Populations
Various nonchemical methods can be used to reduce the growth of mite populations in colonies of honey bees. For example, immigration of mites between apiaries can be limited if the distance between apiaries is increased from 1–2 miles to several miles. Similarly, the distance between colonies within an apiary can be increased from a few feet to 20 feet or more to reduce the drift of varroa mites among colonies. Placing colonies in irregular groups rather than straight rows will also reduce the drift of bees and mites between colonies.
Beekeeping practices may need to be changed to avoid enhancing mite populations. For example, beekeepers commonly combine weakened colonies with stronger ones to save the hive equipment (for example, to keep combs from being damaged by larvae of the small hive beetle). This practice may actually transfer varroa mites and viral diseases to the stronger colony. Evaluate your beekeeping routines and decide if any procedures need modification to avoid inadvertently worsening the varroa mite problem within the apiary.
Other strategies to combat varroa mites include using dusts such as powdered sugar to remove mites from all adult bees in a colony, and purposely creating a break in brood production by caging queen honey bees. Although these methods may be useful under some conditions, they are relatively cumbersome to implement and may have limited effectiveness. The following nonchemical methods are popularly used by beekeepers or have been found to control varroa mite populations by multiple researchers.
Trapping Mites in Drone Brood
The following methods take advantage of the natural preference that varroa mites have for drone brood over worker brood. They prefer drone brood about nine times more than worker brood, and because of the longer development time for drones (24 days) versus workers (21 days), each female mite can produce more mature daughters from drone brood. The strategy is to encourage drone production during periods of the year when colonies typically raise large numbers of drones, such as the spring and early autumn.
The crux of the methods is to allow mites to invade combs containing drone larvae and to then remove the mite-infested drone brood after it is capped and before any adult drones emerge. This point is critical—allowing drones to emerge with varroa mites will increase the resident mite population at a rate much faster than if there were no drone brood. Several researchers have shown mite populations in October to be five to 10 times lower in colonies subjected to drone trapping than in control colonies, and in many cases, the mite populations remained well below the economic or action threshold, and colonies did not need a chemical treatment.
Method 1: Cutting sections of combs that bees rebuild with drone-sized cells.
This method has been popular in Europe for many years. To use the method, combs must be built from wax foundation that is not wire-reinforced or backed by plastic sheets. In the early spring, a narrow section of comb is cut away from the bottom of an already drawn comb. During honey flows, bees tend to rebuild these areas with drone-sized cells (Figure 7). Once built, the queen will lay eggs in the section of drone comb, and varroa mites will invade the drone larvae before the adult bees cap them. Cut away the mite-infested drone brood using a hive tool within 3 weeks after the first drone cells are capped. Destroy the removed strip of drone brood by burning it or placing it into a sealed trash bag. Reinsert the comb into the broodnest, and if the honey flow is strong, two to three cycles of drone trapping can be completed in the spring months.

Method 2: Using commercial drone combs.
Several beekeeping supply companies now sell plastic drone foundation. Sometimes these combs are colored green so that it is easy to find the drone comb when examining a hive. It is best to have at least four drawn drone combs available per colony that you want to treat using this method. Install two of these drawn combs into the active broodnest of a colony in the spring when colony growth is high. It is best to put these combs just inside the last comb of worker brood on either side of the broodnest (Figure 8).
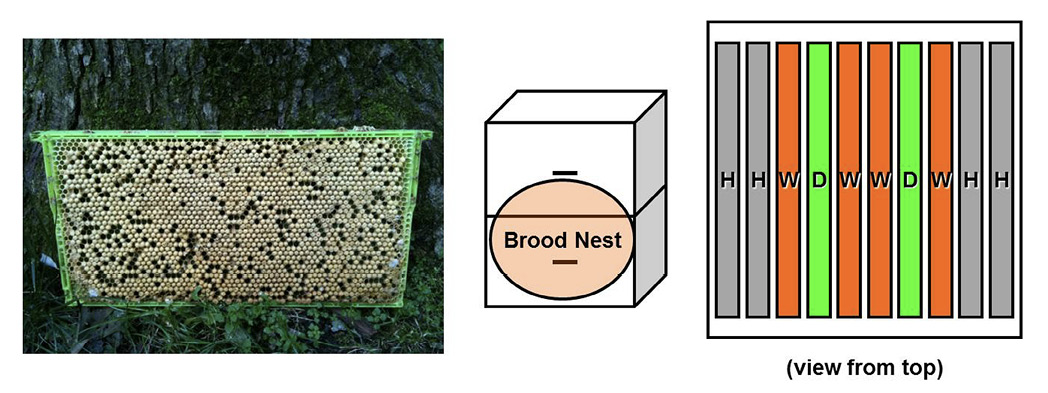
The queen will lay eggs in one or both of the drone combs during periods of major growth in the spring. Varroa mites will invade many of these drones’ cells (perhaps hundreds of them), and the bees will eventually cap the cells. As in the previous method, the important action is to remove capped drone brood before any adult drones emerge from their brood cells. Therefore, it is best to insert and remove the drone combs on a 3-week cycle to stay within the 24 days drones need to develop from egg to adult. The second set of drone combs are installed when the first mite-infested set of combs are removed. This continual supply of empty drone comb will maximize the number of mites that will ultimately be removed from the nest. These mite-infested combs should be frozen for five to seven days to make sure that the brood and mites in the combs are frozen solid. It is not advisable to give the dead drone brood to strong colonies for cleaning out the comb because of the possibility of viruses being transmitted into the colony as hygienic bees eat the dead pupae. As an alternative, fire ants (or other ants) can be encouraged to clean out the combs before reusing them in a hive.
Using Screen Floors
Some research has shown a reduction in varroa mite populations when screened bottom boards are used instead of solid wooden floors in hives. For several studies, the degree of mite reduction was modest: colonies with screened floors had 10–15 percent lower mite populations than did those with solid floors after a couple of months of testing. However, not every study has shown differences between the two types of floors. When differences are found between screened and solid floors, it is not clear if the mites are dropping through the screened floor, or if changes in colony ventilation somehow influence the growth of the mite population. Most researchers agree that screened floors probably do not harm colonies. Screened floors may actually help control other pests such as the small hive beetle (Aethina tumida) by allowing debris that may be attractive to the beetles to fall out of the colony. Additionally, screened floors provide a simple means for sampling for varroa mites by inserting sticky boards (see above).
Using Mite-Resistant Bee Stock
Researchers and bee breeders continue to develop honey bees that resist diseases and parasites. Although much more needs to be done in this area of bee genetics, some honey bees with useful resistance to varroa mites have been developed. Selection for disease resistance can be more difficult than selection of other colony-level traits, such as honey production or defense behavior, because it is difficult to monitor for disease pathogens or pest populations. However, new molecular diagnostic techniques that measure DNA, RNA, and proteins may soon allow the direct selection of genes or genetic markers associated with the genes that control disease resistance in honey bees. Several types of honey bees are currently available from commercial sources. Although varroa resistance in all of these types of bees is not sufficient (if no other IPM techniques are used) to eliminate the need for miticides, research has shown a significant reduction in the number of treatments needed to control mites when varroa-resistant stocks of bees are used in stationary apiaries and migratory beekeeping operations.
Minnesota Hygienic Stock or Stocks Selected for General Hygiene
Many bee breeders select for high nest cleaning or hygienic behavior. Research in the 1940s and 1950s by Walter Rothenbuhler at the Ohio State University indicated that stocks of bees expressing high hygienic behavior had significant resistance to American foulbrood. Dr. Marla Spivak at the University of Minnesota extended this initial work and selected for high general hygiene from existing commercial Italian stocks (Boecking and Spivak 1999). Her selection methods involved using liquid nitrogen to freeze circular sections of capped brood and returning them to bee colonies. Highly hygienic colonies remove more than 90 percent of the freeze-killed bee pupae within 24 hours, while less hygienic colonies remove considerable fewer of the freeze-killed pupae. Bees selected for high rates of removal became known as Minnesota hygienic stock. Research has shown that these bees are significantly resistant to American foulbrood (bacterial disease), chalkbrood (fungal disease), and varroa mites.
ARS Russian Honey Bees
Researchers at the U.S. Department of Agriculture–Agriculture Research Service honey bee breeding, genetics, and physiology laboratory in Baton Rouge, Louisiana, produced a stock of varroa-resistant honey bees that were originally derived from several hundred queen honey bees that were imported from the Primorsky region of far-eastern Russia (Rinderer et al. 2010). It is believed that the original stock represents a population of Western honey bees that probably had the longest exposure to varroa mites because this is an area where the Eastern honey bee (original host of varroa mites) and our Western honey bee have coexisted for about 150 years. The researchers have gone through many generations of selection, and the bees have significant resistance to varroa mites. The stock is currently being sold as ARS Russian Honey Bees by the Russian Queen Breeders Association (http://www.russianbreeder.org/).
Bees Expressing the Varroa Sensitive Hygiene Trait
USDA–ARS researchers produced a second type of varroa-resistant honey bee by selecting for colonies that did not grow mite populations as fast as unselected control stocks. After several years of selection, lines of honey bees were produced in which growth of mite populations were very low, and in the most highly selected lines, mite populations actually decreased over time.
After many years of research, it was discovered that the mechanism of resistance in these bees was varroa sensitive hygiene (VSH) (Rinderer et al. 2010). Adult bees in the resistant colonies can smell mite-infested pupae. When they discover them, the bees uncap the brood cells, remove the mite-infested pupa and discard it from the nest (Figure 9). Although this procedure kills the host bee, it also effectively interrupts the reproductive cycle of the adult female mite. Several of these interruptions will severely reduce the lifetime reproductive output of targeted varroa mites.
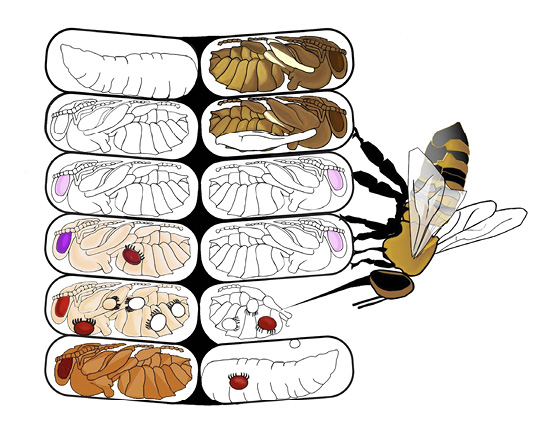
Although VSH is a type of nest-cleaning behavior, it is genetically distinct from general hygiene produced by selecting for the ability to remove freeze-killed brood (see above). Recent tests showed that VSH bees were very good at removing freeze-killed brood and mite-infested pupae, whereas Minnesota hygienic bees were very good at removing freeze-killed brood but much poorer at removing mite-infested pupae from brood cells.
Many commercial suppliers of VSH breeder queens can now be found with an internet search. VSH breeder queens are purebred for the VSH trait. These breeder queens are used to produce daughter queens that are outcrossed to drones from unrelated and nonresistant stocks. There are many commercial queen producers who sell these outcrossed VSH queens for use in standard production colonies (check the advertisements in the bee journals).
Overview of Chemical Treatments
Even when nonchemical methods and resistant stocks of honey bees are used, there may eventually come a time when varroa mite populations exceed the treatment threshold in colonies. When this happens, chemical miticides become the last line of defense. Always think of using chemicals in hives as the last resort. The reasoning is fairly simple. Many of the chemicals used to control varroa mites can have negative health impacts on honey bees (Table 3).
|
Product |
Active ingredient |
Mode of action |
Formulation |
Health effects on bees |
Residue time in combs |
|
Apistan |
tau-fluvalinate |
poisons voltage-gated sodium ion channels in nerves |
impregnated strips |
yes |
years |
|
CheckMite+ |
coumaphos |
poisons the enzyme acetyl-cholinesterase in nerves |
impregnated strips |
yes |
years |
|
Apivar |
amitraz |
binds to octopamine receptors in nerves |
impregnated strips |
yes |
short |
|
Api Life Var |
thymol |
binds receptors in nerves |
solid wafer |
yes |
short |
|
Apiguard |
thymol |
binds GABA receptors in nerves |
slow-release gel |
yes |
short |
|
Mite Away Quick Strips |
formic acid |
strong acid that disrupts electron transport in mitochondria of cells |
soaked onto pads that release fumes |
yes |
short |
|
Formic Pro |
formic acid |
strong acid that disrupts electron transport in mitochondria of cells |
soaked onto pads that release fumes |
yes |
short |
|
HopGuard II |
beta acids from hops |
unknown |
impregnated strips |
unknown |
short |
|
Api-Bioxal |
oxalic acid |
strong acid that disrupts electron transport in mitochondria of cells |
used as a dribble in syrup or as a fumigant |
yes |
short |
In addition, many of these chemicals contaminate combs, and over time, these hive residues could impact colony health. Some of the chemicals remain in combs as residues for years, while others break down in weeks to months. Additionally, mite populations can become genetically resistant to chemicals. Therefore, it is highly recommended that you use chemicals only when the mite population reaches the action threshold, and that you rotate different chemicals every couple of years to slow the development of resistance.
The following guidelines are essential for providing the best protection to honey bees when you use chemicals to control mites:
- Only use chemical treatments when mite populations reach or exceed the economic or treatment threshold.
- Only use chemicals that are legally registered (Section 18 or Section 3) with the Environmental Protection Agency to control varroa mites.
- Always follow the label instructions for each miticide, which includes the proper use of personal protective equipment during the application process and proper disposal of materials after the treatment period.
- Under no circumstances should experimental, homemade, or nonregistered chemicals be applied to hives to control varroa mites (or any other hive disease or pest). This practice is illegal and may result in death of the bee colony, contamination of honey and wax, and severe harm to the beekeeper.
- Rotate miticides every couple of years to slow the development of genetic resistance to any one chemical. It is also a good strategy to use different chemicals within the same year if chemical treatments are warranted in the spring and autumn.
It is important to consider the timing of chemical treatments. Do not apply chemical treatments during honey flows to avoid contamination of honey. The exceptions are Mite Away Quick Strips, Formic Pro, Api-Bioxal, and HopGuard II, which can be legally used during a honey flow. Additionally, do not use chemicals in a colony that is producing winter bees because this could affect the ability of the colony to survive the winter. Also, avoid high varroa mite populations and viral loads during winter bee production to keep the winter bees as healthy as possible. Organic acids (oxalic acid and formic acid) work best when colonies no longer have capped brood, which is typically November into December (Figure 10).
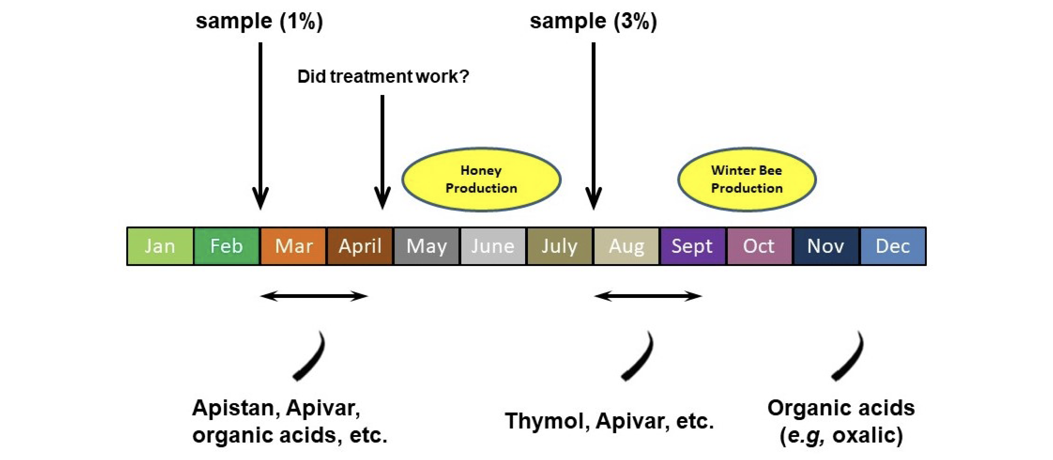
Beekeepers in Mississippi should sample their colonies at least twice a year—once in late February and again in July. Many of the chemical treatments consist of impregnated strips that are inserted into the broodnest for 40–50 days to kill mites. These two sampling periods occur well enough in advance of major honey production or of winter bee production to allow the use of chemical treatments without overlapping with these two important periods of the annual cycle of a colony of bees.
Although there is no published action or economic threshold for February in our area, it is highly recommended that beekeepers sample their colonies at this time. Although mite populations are likely low (many varroa mites die during the winter), the period of most rapid honey bee colony growth occurs in March to May. Populations of varroa mites grow fastest when brood production is highest in the colony.
Treat colonies if the mite load on adult bees exceeds 1 percent in February and if you do not intend to use nonchemical methods to slow the growth of mite populations. That level represents about 500–600 mites in a colony of 15,000–20,000 bees. This level is not damaging, but if left unchecked, the mite population will likely reach the economic threshold within 10 weeks, which will be at the beginning of the major honey flow. Avoid chemical treatment after a February sampling if you intend to use drone trapping methods during March to May (highly recommended).
Finally, consider sampling after a chemical treatment (especially in the spring) to make sure the treatment worked. Various populations of mites have become genetically resistant to chemicals, so they can no longer be killed by these miticides. Some populations have become resistant to more than one chemical. These chemically resistant mites can drift from beekeeper to beekeeper. Rather than trust that a chemical treatment worked, it is best to sample. If the mite load has not been lowered, use a different chemical to control them. The chosen chemical should have a different mode of action than the original chemical.
References
Boecking, O. and M. Spivak. 1999. Behavioral defenses of honey bees against Varroa jacobsoni Oud. Apidologie. 30: 141-158.
Delaplane, K. and M. Hood. 1999. Economic threshold for Varroa jacobsoni Oud. In the southeastern USA. Apidologie 30: 383-395.
Honey Bee Health Coalition. 2022. Tools for Varroa management: A Guide for Effective Varroa Sampling & Control, 8th Edition, The Keystone Policy Center, 37 pages.
Lee, K. V., R. D. Moon, E. C. Burkness, W. D. Hutchison, and M. Spivak. 2010. Practical sampling for Varroa destructor (Acari: Varroidae) in Apis mellfera (Hymenoptera: Apidae) colonies and apiaries. Journal of Economic Entomology. 103 (4): 1039-1050.
Rinderer, T. E., J. W. Harris, G. J. Hunt, and L. I. de Guzman. 2010. Breeding for resistance to Varroa destructor in North America. Apidologie. 41 (3): 409-424.
The information given here is for educational purposes only. References to commercial products, trade names, or suppliers are made with the understanding that no endorsement is implied and that no discrimination against other products or suppliers is intended.
Publication 2826 (POD-08-23)
By Jeff Harris, PhD, Associate Extension/Research Professor, Biochemistry, Molecular Biology, Entomology, and Plant Pathology, Audrey B. Sheridan, Extension/Research Associate II, Biochemistry, Molecular Biology, Entomology, and Plant Pathology, and Joseph A. MacGown, Research Technician/Scientific Illustrator, Biochemistry, Molecular Biology, Entomology, and Plant Pathology.
The Mississippi State University Extension Service is working to ensure all web content is accessible to all users. If you need assistance accessing any of our content, please email the webteam or call 662-325-2262.



